It becomes totally useless when we move down to the subatomic level and consider the lightest of all chemically-significant particles, the electron. key,chapter 14 chemistry study guide answers,chapter 16 emt quiz answers answer key. Choose from a bold, colour-coded version, emphasising readability and clarity, and a visual version with fascinating element artwork. Once we get down to the atomic level, this simple view begins to break down. The table below shows the properties of metals compared to non-metals. Get two for the price of one with this double-sided periodic table poster. 5.3: Light, Particles, and Waves Our intuitive view of the "real world" is one in which objects have definite masses, sizes, locations and velocities. The figure most closely associated with the periodic table is Dmitri Mendeleev, the Russian chemist who devised his first table in 1869.5.2: Quanta - A New View of the World The fact is, however, that it is not only for real, but serves as the key that unlocks even some of the simplest aspects of modern Chemistry. Our goal in this lesson is to introduce you to this new reality, and to provide you with a conceptual understanding of it that will make Chemistry a more meaningful part of your own personal world.5.1: Primer on Quantum Theory A quantum catechism: elementary introduction to quantum theory in the form of a question-and-answer "primer", emphasizing the concepts with a minimum of mathematics.They are good conductors of heat and electricity. 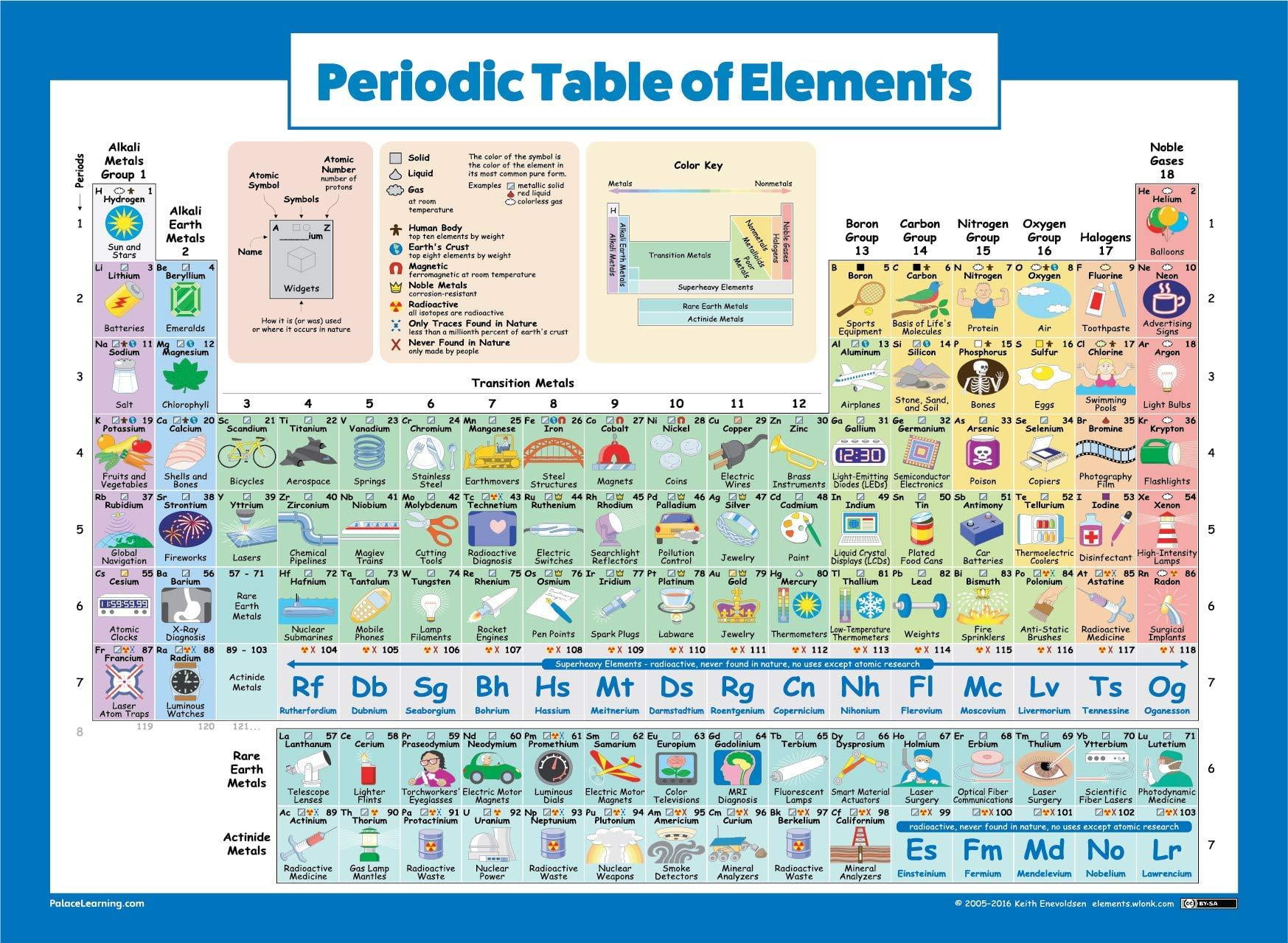
They are also malleable and can be ductile. Metals are solid at room temperature, except for mercury, which is liquid. \)Įverything you need to know in a first-year college course about the principal concepts of quantum theory as applied to the atom, and how this determines the organization of the periodic table. Except for hydrogen, elements on the left side of the periodic table are metals. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
